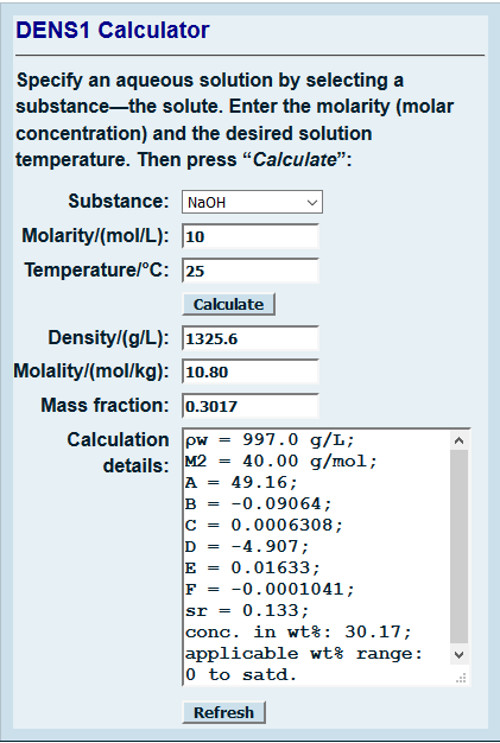
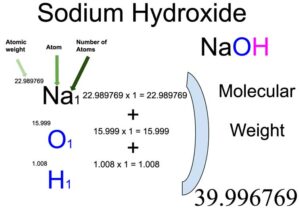
OneClass: Can you please help me make sure the Molarity of NaOH based onmy answers from the final-ini...

Calculate molarity of NaOH in a solution made by mixing 2 L of `1.5 M NaOH,3 L` of 2 M NaOH and 1L - YouTube

1 a. Calculate the number of moles of bne sodium hydroxide in 16.0g of sodium hydroxide (NaOH).b. Calculate - Brainly.in

How to Calculate Analyte Concentration Using the Equivalence Point in an Acid-base Titration | Chemistry | Study.com

SOLVED: Preparation of 0.5 M NaOH solution by dilution Volume of NaOH stock solution (mL) 25.0m| 30 ^ B. Concentration of dilute NaOH solution (M) Calculation of theoretical molarity of the dilute

Calculate the molarity of NaOH in the solution prepared by dissolving its 4 g in enough water - YouTube



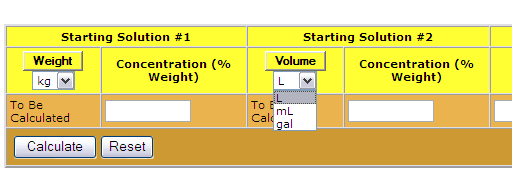

:max_bytes(150000):strip_icc()/how-to-calculate-normality-609580final2-0d5efa5a961f4fa0a7efc780921faee1.png)