Friday Worksheet Name: ………………. Revision 3 1) The 1HNMR spectrum of a compound with the chemical formula C3H6O2

Revisiting the Determination of Percent Aspirin Lab: Using a Limiting Reactant Approach for Students To Also Determine the Amount of Iron(III) Chloride | Journal of Chemical Education

Draw the structure of aspirin. Should this compound test positive with 1% iron (III) chloride solution? Explain. | Homework.Study.com

Low-dose aspirin has limited benefits in adults without heart disease: Yale experts explain why talking to a doctor can help < Yale School of Medicine

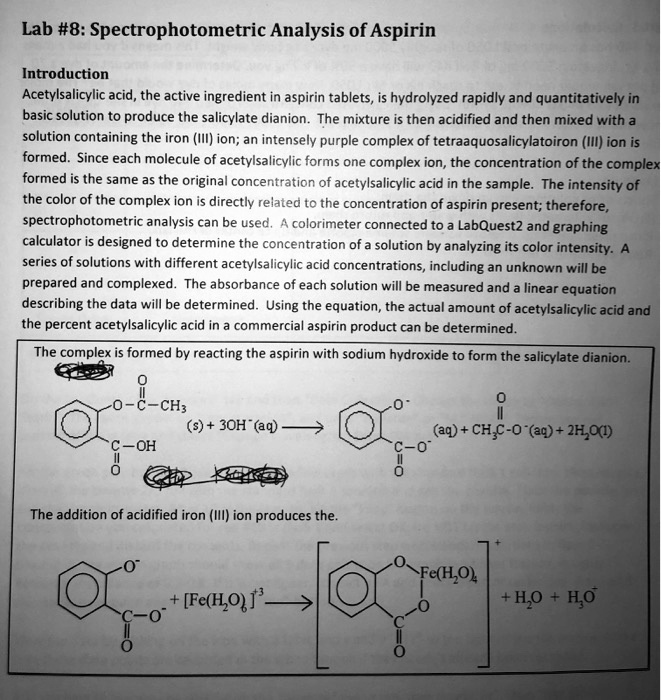
S PECTROPHOTOMETRIC A NALYSIS OF A SPIRIN. Introduction: A colored complex is formed between aspirin and the iron (III) ion. The intensity of the. - ppt download
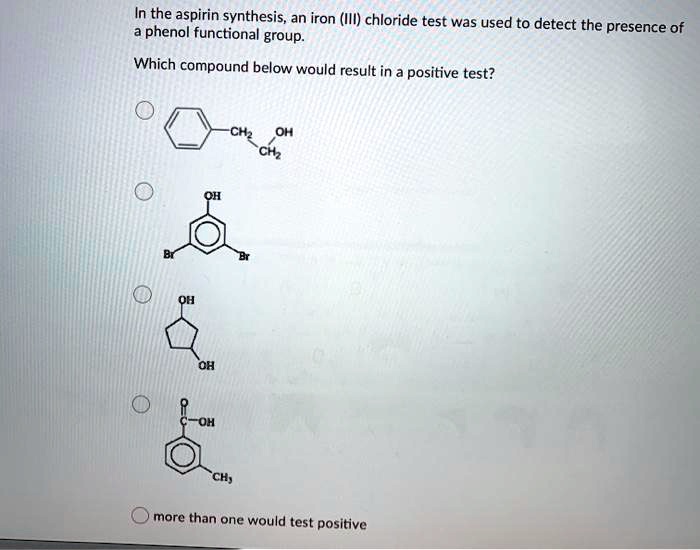
SOLVED: In the aspirin synthesis, an iron (III) chloride test was used to detect the phenol functional group. presence of Which compound below would result in a positive test? CHz Ciz CH;

If color change were to occur after addition of iron (III) chloride to the synthesized aspirin, what would that indicate about the reaction? | Homework.Study.com

PDF) Iron chelation as a possible mechanism for aspirin induced malodealdehyde production by mouse liver microsomes and mitochondria

Does anyone know how to make pure ferric salicylate, I did the ferric chloride test on some expired aspirin and it was the most intense violet-blue I've ever seen, my camera couldn't

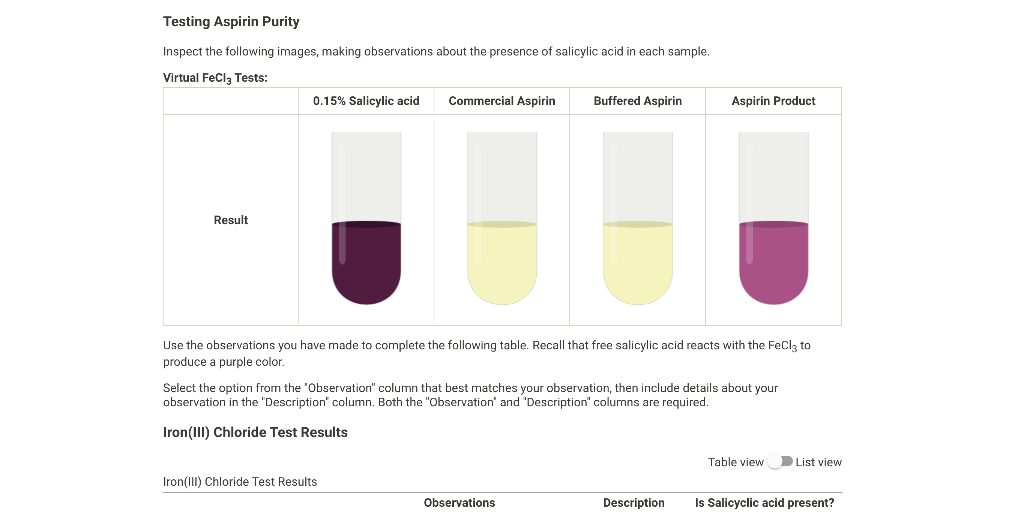
SOLVED: The purity of the aspirin sample is tested with iron (IIl) chloride A purple color resulting from iron (III) chloride test indicates the aspirin sample contains some unreacted salicylic acid and